Showroom
Industrial Coverall is provided by our company
which act as a protective gear designed to safeguard the health of workers by
minimizing the exposure to a biological agent. This coverall will protect the
user against health or safety risks at work.This is very effective as well as
economical.
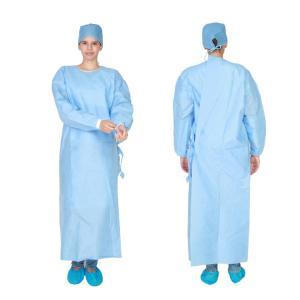
Hospital Uniform offered by us, refers to that
uniform which is designed by our professionals, for the workers and staffs of
hospitals, nursing homes, and clinics. This uniform helps in differentiating
them among others. This uniform offers soft feel, and durable to use. This
uniform is very cost effective and safe for them.
Industrial Cap is very light in weight and
comfortable to wear. This cap is made by utilizing the finest raw materials and
modern machines. This cap is priced reasonably, for maximum client
satisfaction.This cap is very cost effective and can be easily purchased in
bulk quantities.
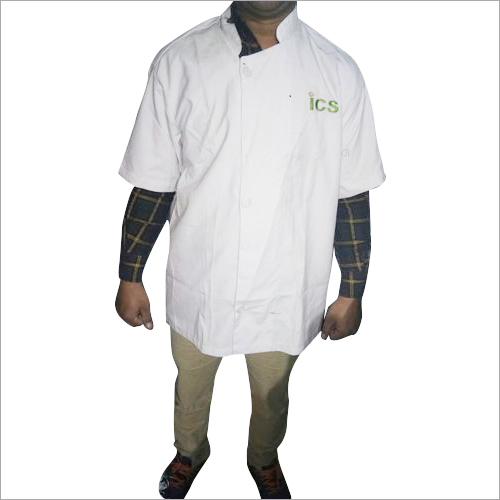
Chef Uniform is designed by our experts using
optimum quality clothing. This uniform helps in protecting the body parts of
chef while making dishes. This uniform helps in handling excess heat during
using ovens, or gas stoves. This uniform requires very low maintenance costs.
This uniform is simple to clean and easy to use.
Industrial Uniform is made for the workers,
labours and people working in the industry. This uniform resembles the workers
for the particular industry.This uniform is tested under different parameters
to ensure its high quality and effectiveness. This uniform is very easy to wear
and simple to clean.
Anti-static Cloths provided, are specialized clothing
worn by an employee for protection against infectious materials. These cloths
create a barrier between the potential infectious material and the worker. They
can include items such as safety helmets, gloves, eye protection,
high-visibility clothing, safety footwear and safety harnesses.
ESD Garments are mainly used for protecting the
person from ever static damage. These garments are manufactured using optimum
level of raw materials. These garments are used to the clean room & an
excellent anti-static effect, dust-proof performance.hey are very effective and
economical in nature.
Boiler Suit is made using upgraded technology, for
Body Protection from harmful effects and damages. This suit acts act as a protective
shield to employees from different types of injuries and illness. Currently.
This requires very low maintenance and replacement costs, hence very cost
effective in nature.
Disposable Coverall Dangri is manufactured by our
premium quality raw materials and high technology. This product is very easy to
wear and simple to use. This coverall is highly appreciated by our customers,
for its high quality. This coverall is very cost effective and can be easily
availed at nominal pricing.
Casual Tie is made in hard wearing polyester,
which has shine and unique look. This tie is known for its unique features,
like high quality, best designs, unique colours, and excellent durability. This
tie can be fitted with elastic neck pieces. This tie is very effective as well
as economical to use.
SHAGUN ENTERPRISES
GST : 09AFIPA9465L1ZX
GST : 09AFIPA9465L1ZX

















































 Send Inquiry
Send Inquiry Send SMS
Send SMS


